Kinetic data by nonisothermal reaction calorimetry: a model-assisted calorimetric evaluation
Hoffmann, Wilfried, Kang, Ying, Mitchell, John C. 

![(ITEM_4168)_ABSTRACT_IMAGE_op060144jn00001.jpg [thumbnail of (ITEM_4168)_ABSTRACT_IMAGE_op060144jn00001.jpg]](https://gala.gre.ac.uk/4168/1.hassmallThumbnailVersion/%28ITEM_4168%29_ABSTRACT_IMAGE_op060144jn00001.jpg) 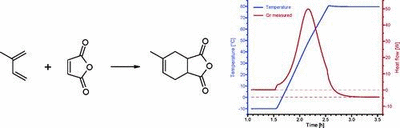 Preview |
Image (JPEG)
(ITEM_4168)_ABSTRACT_IMAGE_op060144jn00001.jpg - Additional Metadata Download (11kB) |
Abstract
The use of a reaction calorimeter in combination with kinetic modeling software to obtain nonisothermal kinetic data is presented. The Diels−Alder reaction of maleic anhydride and isoprene in DMF was used as a model to demonstrate the feasibility of the method. The Arrhenius A factor and the activation energy could be achieved from a single experiment with a reaction calorimeter (Mettler RC1) by fitting the experimental heat generation curve to a second-order kinetic model using commercially available software packages. The use of fitting software revealed a discrepancy between the experimental reaction heat and the heat calculated from the rate parameters. This discrepancy could be resolved by reintegration of the heat generation curve using an adjusted baseline derived from the rate data. The methodology was applied by varying reaction conditions (starting concentrations, heat rate, temperature range), and the results appeared to be independent of these variations within the experimental errors (Ea = 58.5 ± 2.0 kJ mol-1, A factor 4.02 × 106 L mol-1 s-1). It is shown by comparison with literature data that this fast method generates kinetic parameters with a sufficient reliability to be used with process-modeling tools for computer-supported scale-up.
| Item Type: | Article |
|---|---|
| Uncontrolled Keywords: | kinetic data, nonisothermal reaction calorimetry |
| Subjects: | Q Science > QD Chemistry |
| Pre-2014 Departments: | School of Science School of Science > Medway Sciences Research Group |
| Related URLs: | |
| Last Modified: | 16 Apr 2020 13:26 |
| URI: | https://gala.gre.ac.uk/id/eprint/4168 |
Actions (login required)
 |
View Item |
Downloads
Downloads per month over past year
 Tools
Tools Tools
Tools